Conductivity Experiment using Metal
A known property of metal is conduction. Metals are excellent conductors of heat and electricity—heat energy and electrons travel very quickly through them. You can experiment with both heat and electricity conduction using items from around your house.
Adult supervision recommended.
Collect a metal spoon, wooden spoon, and other kitchen implements to compare heat conductivity. Set them in a glass jar of hot, but not boiling, water. Which ones heat up fastest? The ends of the metal utensils should have felt hot first, because they conduct heat better. For a little more excitement, try this activity again using only metal utensils, with a dab of cold butter on top of each utensil. Which one loses its butter first? Why might that be?
Look at each utensil’s handle thickness and length as well as its top surface area (e.g., a wire whisk has less surface to heat the butter than a ladle does) for clues. Also, keep in mind that some metals conduct heat better than others do.
Test an Object for Electric Conductivity
What You Need:
- C- or D-size battery
- Aluminum Foil
- Flashlight or other 1.5- or 3-volt bulb
What You Do:
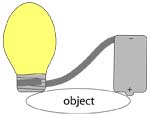
1. Make a long ribbon wire out of the foil by cutting a piece about 18′ x 2′. Fold the foil lengthwise in fourths so that you form a ribbon. Hold or tape one end of this wire to the flat end (negative terminal) of the battery and wrap the other end tightly around the ‘threaded’ (screw) sides of the light bulb.
2. Now you’re ready to test various objects around your house to see if they conduct electricity. Do this by pressing the positive terminal of the battery (the end with a bump) to one side of an object, and the metal end of the light bulb to the other side. If the bulb lights up, a series circuit was formed: electric current can pass unobstructed through the wires from the battery to the light bulb to the battery again.
What are some variables that could keep the light bulb from shining even if the object it was touching was metal? Even though an object may be metal and otherwise a great conductor of electricity, a plastic or paint coating on it could break the circuit connection.
Safety note: Remember never to insert wires or another object into electrical outlets! The electricity generated by the battery for this experiment is in a safe amount, however.
Copper-plating Fun
Try this experiment: first clean pennies, then watch them oxidize and use them to cover (or plate) an iron nail with copper! Although newer pennies contain only a small amount of copper (2.5%), they still have enough for this project.
What You Need:
- Ceramic or plastic bowl
- Vinegar
- Salt
- Pennies
- Paper towels
- Ungalvanized iron nail
1. Fill the bottom of the bowl with vinegar, stir in a teaspoon of salt, and then put 10-15 dull pennies in. Let them sit for five minutes, then take them out and set them on a paper towel to dry. (Don’t dump out the vinegar and salt yet, though!) The pennies will be much shinier than before; this is because vinegar is an acid that ‘eats’ away the oxide layer on the penny that is making it dull. However, if you don’t rinse or dry the clean pennies, after a while you should see a blue layer appear on them. This is a copper oxide compound caused by copper and oxygen reacting with each other; the vinegar (acetic acid) and salt promote the reaction of oxygen with the copper.
2. Now stick an ungalvanized iron nail in the vinegar solution. If you look closely, you will see tiny bubbles along the sides of the nail. Let it sit for 30 minutes and then check to see if there is a dark-brown layer of copper on it. How does that happen? The vinegar solution contains copper from the pennies that it cleaned. When the solution reacts with the nail, it makes a chemical exchange that leaves a copper coating on the nail. When you take the nail out of the solution, the copper will be somewhat sticky; you can set it on a paper towel to dry. Your nail might not be entirely coated, but it will have enough copper on it to see.
You might try this experiment again using only pennies made before 1982 (made with 95% copper).
Did the nail get a copper coating more quickly or in the same amount of time?
Was it a more complete coating?





