Water Properties
One of the things that makes our planet special is the presence of liquid water. Water is fundamental for all life; without it every living thing would die.
Water covers about 70% of Earth’s surface and it makes up 65-75% of our bodies (82% of our blood is water). Even though water might seem boring – no color, taste, or smell – it has amazing properties that make it necessary for supporting life.
(Our need for water and the ocean animals that live in water, is what make oil spills so damaging.)
The chemical composition of water is H2O – two hydrogen atoms and one oxygen atom. The properties of water are special because of the way its atoms bond together to form a water molecule, and the way the molecules interact with each other.
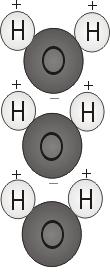
Water Polarity
When the two hydrogen atoms bond with the oxygen, they attach to the top of the molecule rather like Mickey Mouse ears. This molecular structure gives the water molecule polarity, or a lopsided electrical charge that attracts other atoms.
The end of the molecule with the two hydrogen atoms is positively charged. The other end, with the oxygen, is negatively charged. Just like in a magnet, where north poles are attracted to south poles (‘opposites attract’), the positive end of the water molecule will connect with the negative end of other molecules.
What does this mean for us? Water’s polarity allows it to dissolve other polar substances very easily. When a polar substance is put in water, the positive ends of its molecules are attracted to the negative ends of the water molecules, and vice versa. The attractions cause the molecules of the new substance to be mixed uniformly with the water molecules.
Water dissolves more substances than any other liquid – even the strongest acid! Because of this, it is often called the ‘universal solvent.’ The dissolving power of water is very important for life on Earth. Wherever water goes, it carries dissolved chemicals, minerals, and nutrients that are used to support living things.
Because of their polarity, water molecules are strongly attracted to one another, which gives water a high surface tension. The molecules at the surface of the water “stick together” to form a type of ‘skin’ on the water, strong enough to support very light objects. Insects that walk on water are taking advantage of this surface tension.
Surface tension causes water to clump in drops rather than spreading out in a thin layer.
It also allows water to move through plant roots and stems, thermometers, and the smallest blood vessels in your body. As one molecule moves up, it ‘pulls’ the others with it.
Three States of Matter
Water is one of four elements, and the only natural substance that can exist in all three states of matter – solid, liquid, and gas – at the temperatures normally found on Earth.
Many other substances have to be super-heated or -cooled to change states.
The gaseous state of water is present continually in our atmosphere as water vapor. The liquid state is found everywhere in rivers, lakes, and oceans.
The solid state of water, ice, is unique. Most liquids contract as they are cooled, because the molecules move slower and have less energy to resist attraction to each other. When they freeze into solids they form tightly-packed crystals that are much denser than the liquid was originally.
Water doesn’t act this way. When it freezes, it expands: the molecules line up to form a very ‘open’ crystalline structure that is less dense than liquid water.
This is why ice floats. And it’s a good thing it does! If water acted like most other liquids, lakes and rivers would freeze solid and all life in them would die.
Three Other Properties of Water
Water has a high specific heat. That is, unlike air, water can absorb a lot of heat without changing temperature. It’s why swimming feels so refreshing on a hot day.
Water also loses heat slower than air. The result is that in summer, lake and ocean water is usually coldest early in the season and warmest late in the season (after many days of absorbing the sun’s energy).
The boiling point of water (212° F or 100°C at sea level) is affected by altitude. In general, the higher the altitude, the lower the boiling point of water. For example, in Denver, CO (5,280 ft.), water boils at 202.4° F. On Mount Everest in Nepal (29,012 ft.), water boils at 156° F.





