
Make Poinsettia pH Paper
Poinsettia plants practically scream Christmas. So what do you do with them once Christmas is over and the rest of the decor is packed away? Use their red leave for an easy chemistry science project! In this two-part experiment, first you’ll make poinsettia pH paper from poinsettia leaves. Then, you’ll use your poinsettia pH test strips to discover acids and bases.
What You Need:
- 4-5 red poinsettia leaves
- 400 ml beaker
- 250 ml beaker
- Funnel
- Glass petri dish (or other small, shallow dish)
- Heat source, such as a hot plate, lab burner, or alcohol lamp
- Baking rack
- Cookie sheet
- Scissors
- Water
- Filter paper (or coffee filters)
What You Do:
1. Remove 4-5 red leaves from the poinsettia. Use the scissors to cut them into pieces.

2. Place the cut up leaves the bottom of the 400 ml beaker. Add just enough water to cover the leaves.
3. Use your hot plate, lab burner with stand, or alcohol lamp with stand to heat the water to boiling. Be sure to follow lab fire safety protocol! (You may also use a microwave for this step.)

4. Continue simmering a few minutes until the leaves lose their color and the water takes on a deep red tinge. Turn off the heat source. If you’re handling the beaker (removing from the microwave), use caution as the beaker will be hot! Allow the solution to cool.
5. Meanwhile, line the funnel with filter paper. To make a cone, take a piece of filter paper and fold in half, then in half again. Pull one layer open to reveal a cone shape that will fit inside the funnel.

6. Once the beaker is cool enough to touch, carefully pour the liquid through the filter paper-lined funnel into the other beaker. Remove the funnel and discard the remaining plant material and used filter paper.
7. Place another piece of filter paper (or coffee filter) into the petri dish (or other shallow dish).
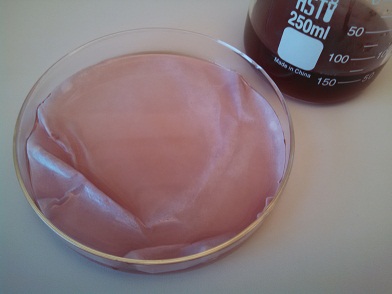
8. Carefully pour the filtered liquid over the paper.
9. Remove the now-saturated filter paper from the shallow dish and allow to dry. Consider laying the saturated paper across a baking rack with paper towels or a cookie sheet underneath.
10. Once your poinsettia pH paper is dry, cut it into strips. Now it’s ready to use!
What Happened:
Like red cabbage (which can also be boiled to make pH test strips), poinsettia leaves contain a chemical pigment called anythocyanin. It’s responsible for giving poinsettia leaves their deep red color. It’s also behind the color of red cabbage, blueberries, and the fall colors of some leaves. Anthocyanin is also pH sensitive, which is why you can use it to make poinsettia pH paper.
Remember that the pH scale allows us to measure how acidic or basic a solution is. Many varieties of pH test strips are commercially available, but some plants contain chemicals that can be used to make your own pH test strips! With these poinsettia pH strips, acids will turn the pigments in the indicator to an orange or reddish color. Bases will turn the pigments in the poinsettia pH strips yellow-green, blue or purple. Neutral substances will show no change on the poinsettia pH paper test strip.
Follow the steps in this project that accompanies the red cabbage pH test strip experiment to use your poinsettia strips to compare solutions for acidity and alkalinity (basicity). And read this article about acids and bases.





